In a study published in
Nucleic Acids Research, a research team led by Prof. ZHANG Kaiming from University of Science and Technology of China (USTC) found the Gp168 protein of bacteriophage Twort inhibited β-clamp function by occupying the DNA sliding channel.
During the process of bacterial DNA replication, DNA Polymerase, which catalyzes the synthesis of DNA, plays an indispensable role. In order to keep DNA Polymerase III sliding along, a protein complex namely β-clamp forms a ring-like structure around DNA, serving as a processivity-promoting factor. Due to the critical function of β-clamp in the replication of bacteria and the formation of tumors, various β-clamp inhibitors have been investigated to a great extent for antibacterial studies.
Previous studies reported that all of the discovered β-clamp inhibitors and binding proteins engaged the β-clamp via hydrophobic protein-binding pocket. In a recent study led by Prof. ZHANG, however, has found that such hydrophobic protein-binding pocket is not a must.
The research team reported a cryo-EM structure of the clamp-Gp168 complex at 3.2-A° resolution. At such high resolution, researchers obtained the structural details of the complex. They found that the Gp168 dimer occupied the DNA sliding channel of β-clamp and blocked its loading onto DNA.
To understand the formation of the Gp168-clamp complex, researchers identified that Gp168 existed as a hexameric complex in solution. The pulled-down experiment involving Gp168 and the β-clamp of Staphylococcus aureus showed that Gp168 could form a stable complex with the β-clamp of Staphylococcus aureus. Researchers then performed the cryo-EM single-particle analysis to further understand the detailed structure of the Gp168-clamp complex.
Based on the results, they concluded that Gp168 represented a new class of β-clamp inhibitor with other members yet to be discovered.
This study is a breakthrough in inhibitory mechanism against β-clamp since the Gp168 protein is the first β-clamp binding molecule that does not target hydrophobic protein-binding pocket. Gp168 protein is also the first natural β-clamp inhibitor produced by bacteriophages discovered so far.
The study not only reveals an alternative mechanism for bacteriophages to inhibit β-clamp but also provides inspiration for the development of new antimicrobial reagents.
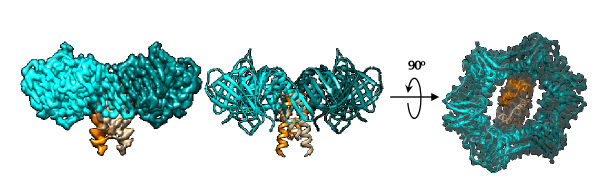
Cryo-EM structure of the clamp-Gp168 complex (Image by LIU Bing et al.)
(Written by ZHANG Yuxin, edited by LI Xiaoxi, USTC News Center)